 Tachycardia does not exceed programmed upper limit rate on pacemaker.Most commonly: paced ventricular beat -> retrograde AV node conduction -> intrinsic P wave -> device reacts to intrinsic P wave by looking for intrinsic QRS, but since AV node is now refractory it delivers a paced beat at the programmed P-R interval (typically ~200-250ms), starting the process anew.Formation of a re-entrant circuit causing inappropriate tachycardia.Potentially life-threatening as it can cause V-Fib or (paradoxically) bradycardia due to failure to capture.Signals generated by interaction of different portions of the pacing system.Physiologic electrical activity (T waves, muscle potentials).New intrinsic arrhythmia (AF has a smaller depolarization than sinus beat), AMI, electrolyte abnormalities, lead separation, battery depletion.Voltages of patient's intrinsic QRS complex is too low to be detected.Failure to sense results in a paced beat on top of an intrinsic beat (as the device is "unaware" of the intrinsic beat").Normal function: a sensed myocardial depolarization greater than the programmed threshold causes inhibition of pacing.Crosstalk - type of oversensing where the ventricular lead senses atrial pacing stimulus, and ventilator output inhibited.Oversensing - most common cause: retrograde P’s, T’s, skeletal muscle myopotentials,.Failure to deliver a stimulus to the heart.Technical - insufficient device output, lead dislodgment, fracture, insulation defect, ventricular wall perforation.Medical - drugs, myocardial disease, electrolytes.Functional - refractory myocardium, desensitized local tissue around the lead.Delivery of pacing stimulus without depolarization.Twiddler Syndrome after large pocket and defibrillator wires coiled around the generator Typically occurs shortly after placement.2% local wound infection 1% sepsis/bacteremia.Unipolar Cautery - can cause sensing and pacing malfunction as well as reprogrammingĭifferential Diagnosis Pacemaker Malfunction Problems with pocket.Cardioversion: Use AP pads >8cm from device to minimize adverse effects.MRI: mostly safe, consult cards on device specific recs.Airport security: may trigger alarm, no alteration of activity.Cell phones: do not interact with device. 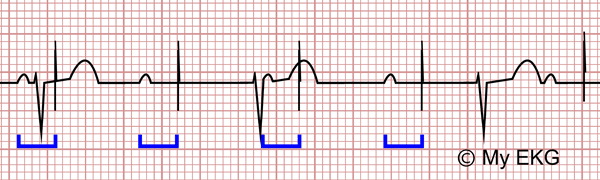
Manufacturer code on pulse generator is visible on Chest Xray.Boston Scientific Inc.: 100 bpm 85 bpm when battery is ready for replacement.Jude Medical Inc.: 98.6 bpm 86.3 bpm when battery is ready for replacement Medtronic Inc.: 85 bpm 65 bpm when battery is ready for replacement.Magnet mode - with placement of a magnet over the device, the mode changes to asynchronous (i.e.(recently acquired by Abbott, Inc.) (1-80) Manufacturer Hotline has patient database.Patient most often has a pocket card indicating manufacturer.Keeps a rate histogram, as well as % of time spent in AF.Keeps track of % of paced vs intrinsic beats.Can record rhythm strips of AF, VT, and VF episodes for later review.Rarely, externally placed electrode during open surgical procedure.Coronary veins along external LV wall via coronary sinus.Left Ventricle (most commonly placed for cardiomyopathy or CHF).Neurocardiogenic Syncope and Carotid Sinus Syndrome.  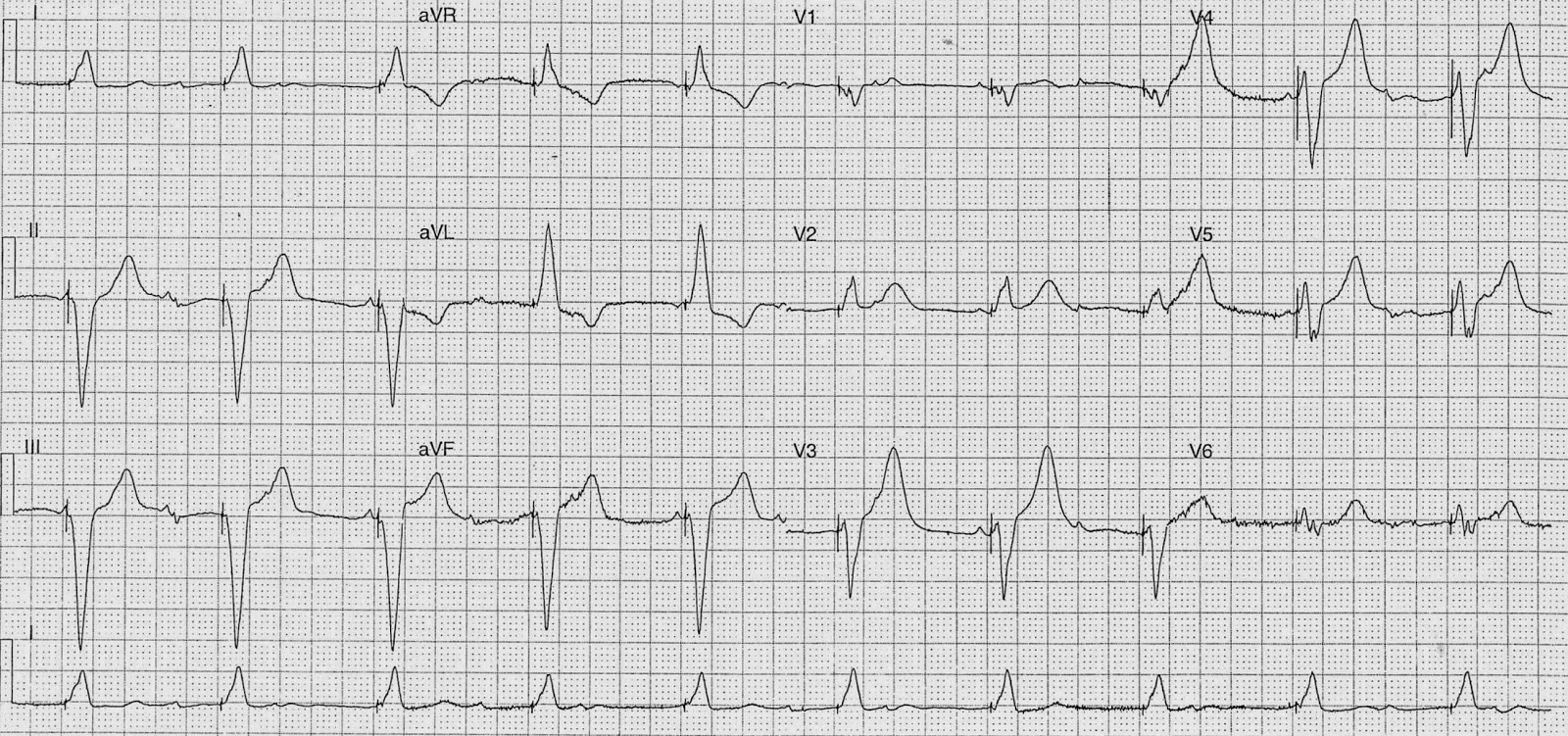
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
